Identifying novel pathways to treat Hirschsprung-associated enterocolitis
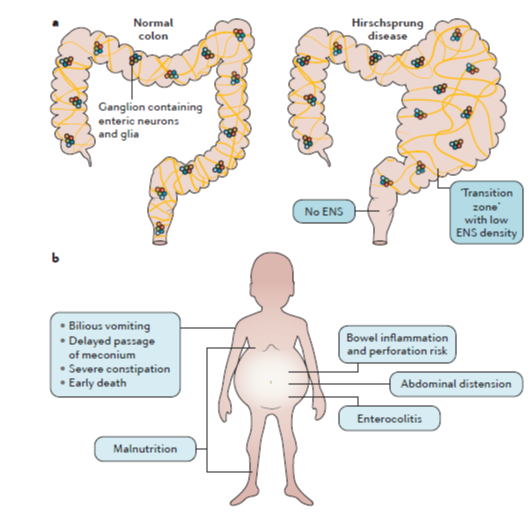
Hirschsprung’s disease (HD) is a common cause of neonatal bowel obstruction. It is characterised by an absence of enteric neurons (aganglionosis) in the distal bowel. The aganglionosis involves the internal anal sphincter and extends orally, most often to the sigmoid colon. The aganglionic segment of the colon fails to relax, causing a functional, severe bowel obstruction. Current treatment is surgical resection of aganglionic bowel. Unfortunately, many children have problems after surgery, particularly in the first years of life. Postoperative problems include constipation, obstructive defecation, fecal incontinence and Hirschsprung-associated enterocolitis (HAEC).
HAEC is a dangerous clinical symptom complex characterized by abdominal distension, fever and diarrhoea. In addition, HAEC has been shown to be associated with reduced functional outcome, particularly fecal incontinence. HAEC occurs in 30–60% of HD patients and may occur both pre-and postoperatively. In some children, HAEC is the presenting symptom of HD, whereas others have one or more episodes after surgery. HAEC is most frequent the first year postoperatively, but some children continue to have relapsing HAEC episodes for many years. HAEC aetiology is poorly understood. Interestingly, this symptom complex does not occur in otherwise healthy children with severe chronic constipation or other bowel disorders. The aim of this project is to increase current understanding of HAEC aetiology with the goal to improve treatment for this condition, which causes severe morbidity and even may be life-threatening.
We hypothesize that the lack of enteric neurons affects the development and function of immune cells in the gut and that disturbances of mucosal immunity in patients with HD is an underlying cause for HAEC.
The aim of this project is to study the mucosal immune system in the gut of HD patients by computational integration of single cell RNA sequencing, Spatial Transcriptomics and high dimensional immune-imaging of colonic tissue from HD patients and age-matched controls.